|
Īnother set of techniques using mass-spectroscopy is often used, but I'm not going to try to explain that here! Note however, this can get very complicated, since there may be multiple copies of one or more identical proteins within a complex, some proteins may be of similar sizes, some proteins may have been broken down into smaller parts, you may have not completely purified your protein. /protein-structure-373563_final11-5c81967f46e0fb00012c667d.png)
The number of different bands corresponds to the number of different proteins. You then stain the gel with something that interacts with all proteins but not the gel. Because different proteins are usually different sizes they move at different rates through the gel. The mixture is then dragged through a gel by applying an electric field. This (usually) results in all the individual proteins being separated and stretched out into negatively charged rods. If you have a purified protein complex a very standard technique involves denaturing the protein in an anionic detergent (SDS). 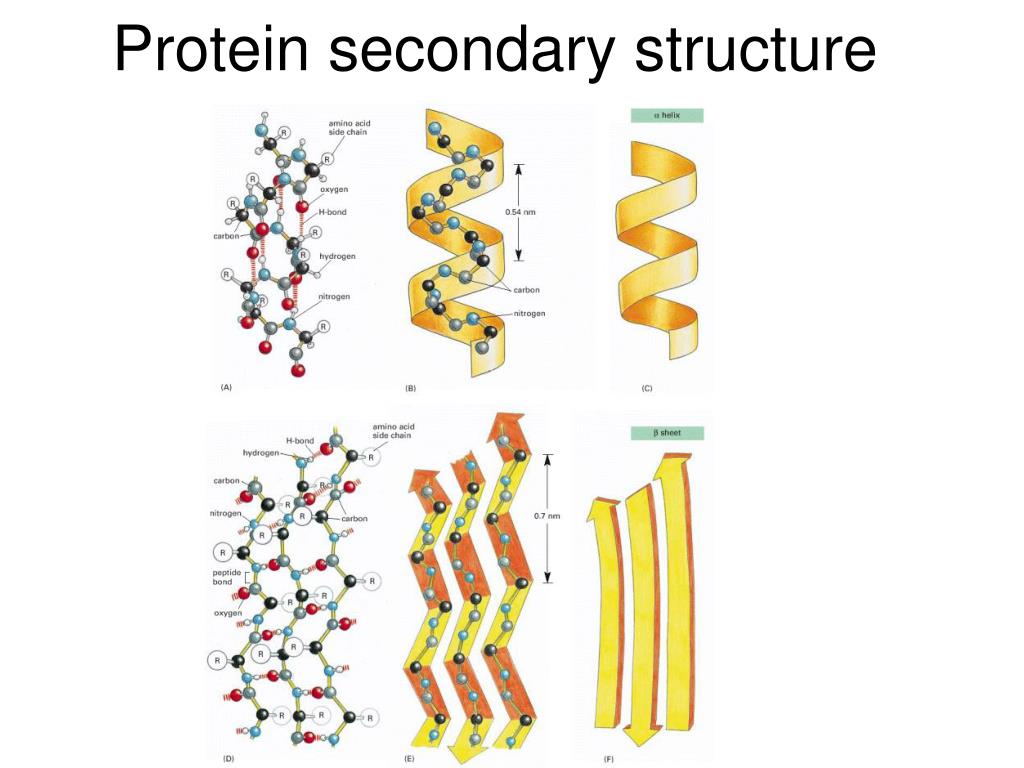
And, note that there is a π bond between the C and O, which makes available additional electron density to O. For example, it matters what else the atoms involved might be bonded to.īut the difference in electronegativity of C and O is 0.89 which is sufficient to polarize a bond. There are many factors that go into determining how polarized a bond is. There are a few other ways it can happen, but it is usually that way.Īs for what you mentioned about the electronegativity difference between O and C, please understand that electronegativity difference is a rule of thumb, not a strict law. Most of the time, the attraction will be between an H and N, O or F. 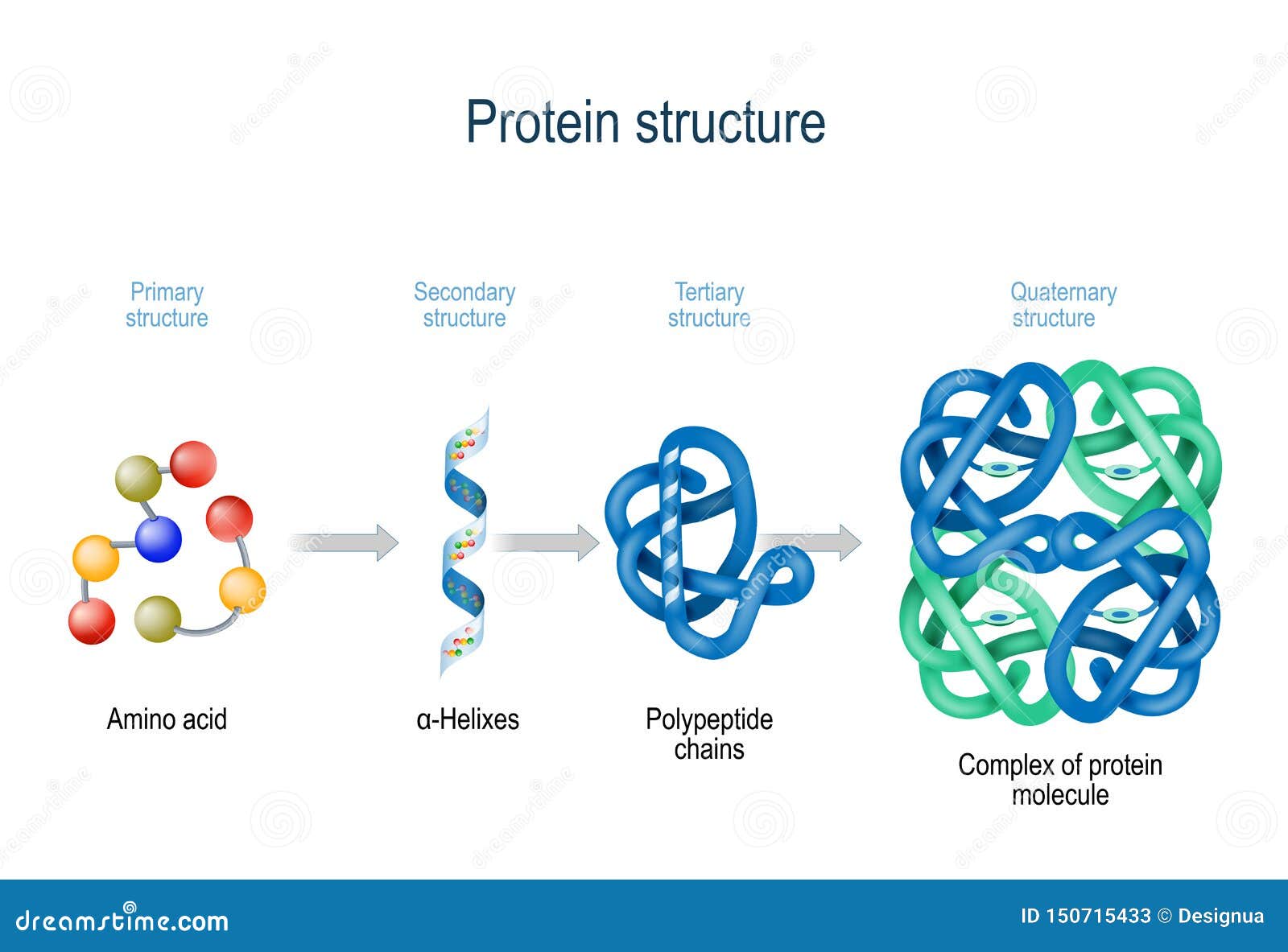
This is not a true bond, just an electrostatic attraction. When, either in separate or the same molecule, you have a partially positively charged hydrogen come close to certain kinds of partially negatively charged groups, then you get an electrostatic attraction between the two groups. You should probably review hydrogen bonding in the chemistry section.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
